The Health Issue: Local innovators help create modern miracles
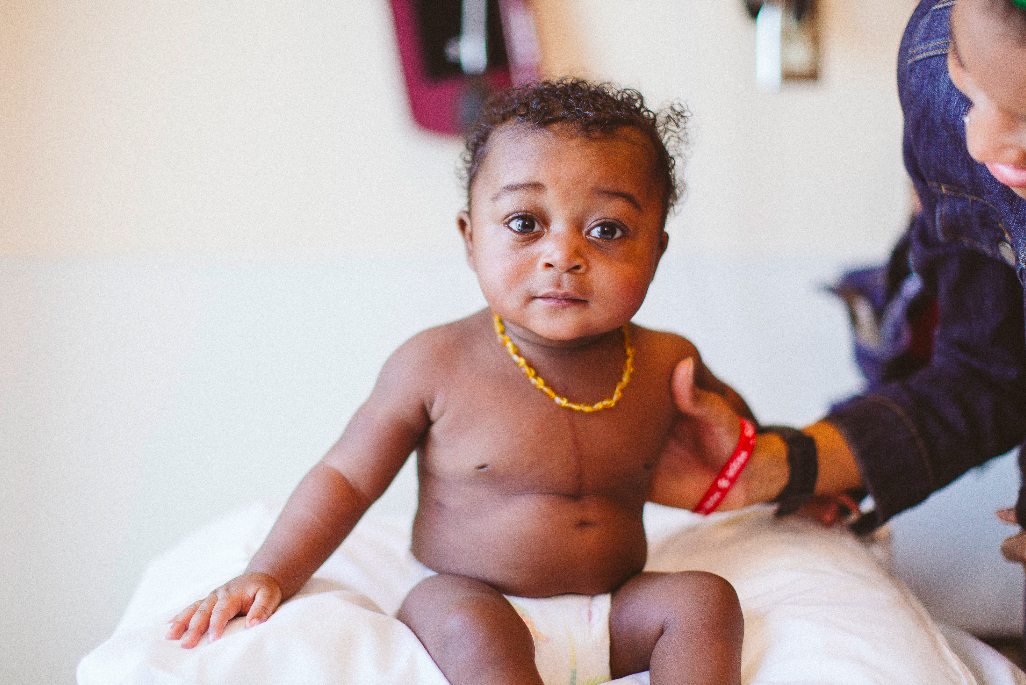
Prince Adewale-Yusuf, 8 months, underwent two heart surgeries before he was 6 months old. His parents used the UVA-developed Building Hope app to stay connected with his health care team during his recovery. Photo by Eze Amos
Support C-VILLE Weekly
C-VILLE Weekly is Charlottesville’s leading newspaper. Founded in 1989, it’s been the area’s local source for informative (and informed) stories in news, arts, and living for more than 26 years.
UP NEXT
In brief
Breaking camp
Shortened stay
Explosive growth
IN CASE YOU MISSED IT
In brief
Breaking camp
Shortened stay
Explosive growth
PUBLICATIONS
C-VILLE Weekly | May 1, 2024
C-VILLE Weekly | April 24, 2024
C-VILLE Weekly | April 17, 2024
C-VILLE Weekly | April 10, 2024
Real Estate Weekly | May 1, 2024
Real Estate Weekly | April 24, 2024
Real Estate Weekly | April 17, 2024
Real Estate Weekly | April 10, 2024
Abode | Spring 2024
Knife & Fork | Spring/Summer 2024
Fine Properties | Spring 2024
Weddings | Fall/Winter 2023
Best of C-VILLE 2023
We Are C-VILLE | March 2024
Summer Camp Guide | March 2024
National Nonprofit Day | August